Снижение массы скелетных мышц при старении в основном объясняется уменьшением размера мышечных волокон II типа
Снижение массы скелетных мышц при старении в основном связано с меньшим размером мышечных волокон II типа. Поэтому увеличение мышечной массы после длительной тренировки с отягощениями может быть полностью связано со специфической гипертрофией мышечных волокон II типа.
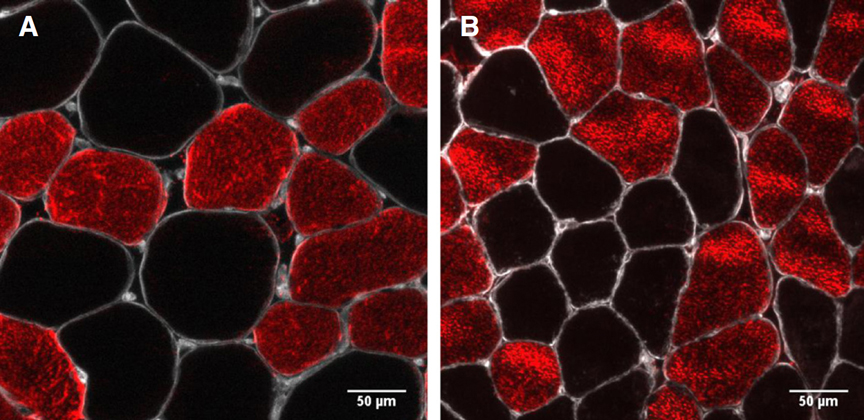
Nilwik R. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size /R. Nilwik , T. Snijders, M. Leenders, B.B.L. Groen, J. van Kranenburg, L. B. Verdijk, L. J.C. van Loon // Experimental Gerontology, 2013. V. 48. P. 492-498.
Р. Нилвик с соавт.
Снижение массы скелетных мышц при старении в основном объясняется уменьшением размера мышечных волокон II типа
История вопроса
Потеря массы скелетных мышц при старении объясняется снижением количества мышечных волокон и размера мышечных волокон.
Цель
Определить, в какой степени различия в площади поперечного сечения мышц ног (CSA) между молодыми и пожилыми мужчинами объясняются различиями в размерах мышечных волокон.
Методы
Измеряли CSA четырехглавой мышцы и размер мышечных волокон типа I и типа II у здоровых молодых (n = 25; 23 ± 1 года) и пожилых (n = 26; 71 ± 1 года) мужчин. Впоследствии пожилые субъекты проводили 6 месяцев тренировочных упражнений с отягощением, после чего измерения повторяли. Различия в CSA четырехглавой мышцы сравнивали с различиями в размерах мышечных волокон I и II типа.
Результаты
CSA четырехглавой мышцы бедра было значительно меньше у пожилых мужчин по сравнению с молодыми (68±2 см2 против 80 ± 2 см2 соответственно; P<0,001). Размер мышечных волокон типа II был значительно меньше у пожилых людей по сравнению с молодыми (29%; P<0,001) с тенденцией к уменьшению мышечных волокон типа I (P = 0,052). Различия в размерах мышечных волокон II типа полностью объясняют различия в CSA четырехглавой мышцы бедра между группами. Длительная тренировка с отягощениями у пожилых людей увеличила размер мышечных волокон II типа на 24 ± 8% (P<0,01), объясняя 100 ± 3% увеличения CSA четырехглавой мышцы (с 68 ± 2 см2 до 74 ± 2 см2).
Вывод
Уменьшение массы скелетных мышц при старении в основном связано с меньшим размером мышечных волокон II типа и, как таковое, вряд ли сопровождается значительной потерей мышечных волокон. Таким образом, увеличение массы скелетных мышц после длительной тренировки с отягощениями может быть полностью связано со специфической гипертрофией мышечных волокон II типа.
Background
The loss of skeletal muscle mass with aging has been attributed to a decline in muscle fiber number and muscle fiber size.
Objective
To define to what extent differences in leg muscle cross-sectional area (CSA) between young and elderly men are attributed to differences in muscle fiber size.
Methods
Quadriceps muscle CSA and type I and type II muscle fiber size were measured in healthy young (n=25; 23± 1y) and older (n=26; 71±1y) men. Subsequently, the older subjects performed 6 months of resistance type exercise training, after which measurements were repeated. Differences in quadriceps muscle CSA were compared with differences in type I and type II muscle fiber size.
Results
Quadriceps CSA was substantially smaller in older versus young men (68± 2 vs 80± 2 cm2, respectively; Pb0.001). Type II muscle fiber size was substantially smaller in the elderly vs the young (29%; Pb0.001), with a tendency of smaller type I muscle fibers (P=0.052). Differences in type II muscle fiber size fully explained differences in quadriceps CSA between groups. Prolonged resistance type exercise training in the elderly increased type II muscle fiber size by 24± 8% (Pb0.01), explaining 100± 3% of the increase in quadriceps muscle CSA (from 68±2 to 74± 2 cm2).
Conclusion
Reduced muscle mass with aging is mainly attributed to smaller type II muscle fiber size and, as such, is unlikely accompanied by substantial muscle fiber loss. In line, the increase in muscle mass following prolonged resistance type exercise training can be attributed entirely to specific type II muscle fiber hypertrophy.
- American Diabetes Association, 2012. Diagnosis and classification of diabetes mellitus. Diabetes Care 35 (Suppl 1), S64–S71.
- Andersen, J.L., Terzis, G., Kryger, A., 1999. Increase in the degree of coexpression of my osin heavy chain isoforms in skeletal muscle fibers of the very old. Muscle Nerve 22, 449–454.
- Brown, M., 1987. Change in fibre size, not number, in ageing skeletal muscle. Age Ageing 16, 244–248.
- Buford, T.W., Anton, S.D., Judge, A.R., Marzetti, E., Wohlgemuth, S.E., Carter, C.S., Leeuwenburgh, C., Pahor, M., Manini, T.M., 2010. Models of accelerated sarcopenia: critical pieces for solving the puzzle of age-related muscle atrophy. Ageing Res. Rev. 9, 369–383.
- Candow, D.G., Forbes, S.C., Little, J.P., Cornish, S.M., Pinkoski, C., Chilibeck, P.D., 2012. Effect of nutritional interventions and resistance exercise on aging muscle mass and strength. Biogerontology 13, 345–358.
- D’Antona, G., Pellegrino, M.A., Adami, R., Rossi, R., Carlizzi, C.N., Canepari, M., Saltin, B., Bottinelli, R., 2003. The effect of ageing and immobilization on structure and function of human skeletal muscle fibres. J. Physiol. 552, 499–511.
- Degens, H., Alway, S.E., 2006. Control of muscle size during disuse, disease, and aging. Int. J. Sports Med. 27, 94–99.
- Dirks, A.J., Leeuwenburgh, C., 2005. The role of apoptosis in age-related skeletal muscle atrophy. Sports Med. 35, 473–483.
- Doherty, T.J., 2003. Invited review: aging and sarcopenia. J. Appl. Physiol. 95, 1717–1727.
- Dreyer, H.C., Blanco, C.E., Sattler, F.R., Schroeder, E.T., Wiswell, R.A., 2006. Satellite cell numbers in young and older men 24 hours after eccentric exercise. Muscle Nerve 33, 242–253.
- Faulkner, J.A., Larkin, L.M., Claflflin, D.R., Brooks, S.V., 2007. Age-related changes in the structure and function of skeletal muscles. Clin. Exp. Pharmacol. Physiol. 34, 1091–1096.
- Fleg, J.L., Lakatta, E.G., 1988. Role of muscle loss in the age-associated reduction in VO2 max. J. Appl. Physiol. 65, 1147–1151.
- Frontera, W.R., Meredith, C.N., O’Reilly, K.P., Knuttgen, H.G., Evans, W.J., 1988. Strength conditioning in older men: skeletal muscle hypertrophy and improved function. J. Appl. Physiol. 64, 1038–1044.
- Goodpaster, B.H., Thaete, F.L., Kelley, D.E., 2000. Composition of skeletal muscle evaluated with computed tomography. Ann. N. Y. Acad. Sci. 904, 18–24.
- Hikida, R.S., Staron, R.S., Hagerman, F.C., Walsh, S., Kaiser, E., Shell, S., Hervey, S., 2000. Effects of high-intensity resistance training on untrained older men. II. Muscle fiber characteristics and nucleo-cytoplasmic relationships. J. Gerontol. A Biol. Sci. Med. Sci. 55, B347–B354.
- Hodson-Tole, E.F., Wakeling, J.M., 2008. Motor unit recruitment patterns 2: the influence of myoelectric intensity and muscle fascicle strain rate. J. Exp. Biol. 211, 1893–1902.
- Janssen, I., Heymsfifield, S.B., Wang, Z.M., Ross, R., 2000. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 89, 81–88.
- Johnston, A.P., De Lisio, M., Parise, G., 2008. Resistance training, sarcopenia, and the mitochondrial theory of aging. Appl. Physiol. Nutr. Metab. 33, 191–199.
- Klein, C.S., Marsh, G.D., Petrella, R.J., Rice, C.L., 2003. Muscle fiber number in the biceps brachii muscle of young and old men. Muscle Nerve 28, 62–68.
- Klitgaard, H., Mantoni, M., Schiaffifino, S., Ausoni, S., Gorza, L., Laurent-Winter, C., Schnohr, P., Saltin, B., 1990. Function, morphology and protein expression of ageing skeletal muscle: a cross-sectional study of elderly men with different training backgrounds. Acta Physiol. Scand. 140, 41–54.
- Koopman, R., 2011. Dietary protein and exercise training in ageing. Proc. Nutr. Soc. 70, 104–113.
- Kosek, D.J., Kim, J.S., Petrella, J.K., Cross, J.M., Bamman, M.M., 2006. Effificacy of 3 days/wk resistance training on myofiber hypertrophy and myogenic mechanisms in young vs. older adults. J. Appl. Physiol. 101, 531–544.
- Kraemer, W., Fry, A., 1995. Strength testing: development and evaluation of methodology. In: Maud, P., Foster, C. (Eds.), Physiological Assessment of Physical Fitness. Human Kinetics, Leeds.
- Kryger, A.I., Andersen, J.L., 2007. Resistance training in the oldest old: consequences for muscle strength, fiber types, fiber size, and MHC isoforms. Scand. J. Med. Sci. Sports 17, 422–430.
- Kuipers, H., Peeze Binkhorst, F.M., Hartgens, F., Wijnen, J.A., Keizer, H.A., 1993. Muscle ultrastructure after strength training with placebo or anabolic steroid. Can. J. Appl. Physiol. 18, 189–196.
- Lang, T., Streeper, T., Cawthon, P., Baldwin, K., Taaffe, D.R., Harris, T.B., 2010. Sarcopenia: etiology, clinical consequences, intervention, and assessment. Osteoporos. Int. 21, 543–559.
- Larsson, L., Sjodin, B., Karlsson, J., 1978. Histochemical and biochemical changes in human skeletal muscle with age in sedentary males, age 22–65 years. Acta Physiol. Scand. 103, 31–39.
- Leenders, M., Verdijk, L.B., van Der Hoeven, L., van Kranenburg, J., Nilwik, R., Wodzig, W.K., Senden, J.M., Keizer, H.A., van Loon, L.J., 2013. Protein supplementation during resistance-type exercise training in the elderly. Med. Sci. Sports Exerc. 45 (3), 542–552.
- Leenders, M., Verdijk, L.B., van der Hoeven, L., van Kranenburg, J., Nilwik, R., van Loon, L.J., in press. Elderly men and women benefit equally from prolonged resistance-type exercise training. J. Gerontol. A Biol. Sci. Med. Sci.
- Lemmer, J.T., Hurlbut, D.E., Martel, G.F., Tracy, B.L., Ivey, F.M., Metter, E.J., Fozard, J.L., Fleg, J.L., Hurley, B.F., 2000. Age and gender responses to strength training and detraining. Med. Sci. Sports Exerc. 32, 1505–1512.
- Lexell, J., Taylor, C.C., Sjostrom, M., 1988. What is the cause of the ageing atrophy? Total number, size and proportion of different fifiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 84, 275–294.
- Macaluso, A., De Vito, G., 2004. Muscle strength, power and adaptations to resistance training in older people. Eur. J. Appl. Physiol. 91, 450–472.
- Mackey, A.L., Kjaer, M., Charififi, N., Henriksson, J., Bojsen-Moller, J., Holm, L., Kadi, F., 2009. Assessment of satellite cell number and activity status in human skeletal muscle biopsies. Muscle Nerve 40, 455–465.
- Martel, G.F., Roth, S.M., Ivey, F.M., Lemmer, J.T., Tracy, B.L., Hurlbut, D.E., Metter, E.J., Hurley, B.F., Rogers, M.A., 2006. Age and sex affect human muscle fibre adaptations to heavy-resistance strength training. Exp. Physiol. 91, 457–464.
- Marzetti, E., Calvani, R., Bernabei, R., Leeuwenburgh, C., 2012. Apoptosis in skeletal myocytes: a potential target for interventions against sarcopenia and physical frailty — a mini-review. Gerontology 58, 99–106.
- Mayhew, J.L., Prinster, J.L., Ware, J.S., Zimmer, D.L., Arabas, J.R., Bemben, M.G., 1995. Muscular endurance repetitions to predict bench press strength in men of different training levels. J. Sports Med. Phys. Fitness 35, 108–113.
- McCall, G.E., Byrnes, W.C., Dickinson, A., Pattany, P.M., Fleck, S.J., 1996. Muscle fiber hypertrophy, hyperplasia, and capillary density in college men after resistance training. J. Appl. Physiol. 81, 2004–2012.
- Narici, M.V., Reeves, N.D., Morse, C.I., Maganaris, C.N., 2004. Muscular adaptations to resistance exercise in the elderly. J. Musculoskelet. Neuronal. Interact. 4, 161–164.
- Peterson, M.D., Rhea, M.R., Sen, A., Gordon, P.M., 2010. Resistance exercise for muscular strength in older adults: a meta-analysis. Ageing Res. Rev. 9, 226–237.
- Roth, S.M., Ivey, F.M., Martel, G.F., Lemmer, J.T., Hurlbut, D.E., Siegel, E.L., Metter, E.J., Fleg, J.L., Fozard, J.L., Kostek, M.C., Wernick, D.M., Hurley, B.F., 2001. Muscle size responses to strength training in young and older men and women. J. Am. Geriatr. Soc. 49, 1428–1433.
- Sheard, P.W., Anderson, R.D., 2012. Age-related loss of muscle fifibres is highly variable amongst mouse skeletal muscles. Biogerontology 13, 157–167.
- Snijders, T., Verdijk, L.B., van Loon, L.J., 2009. The impact of sarcopenia and exercise training on skeletal muscle satellite cells. Ageing Res. Rev. 8, 328–338.
- Snijders, T., Verdijk, L.B., Beelen, M., McKay, B.R., Parise, G., Kadi, F., van Loon, L.J., 2012. A single bout of exercise activates skeletal muscle satellite cells during subsequent overnight recovery. Exp. Physiol. 97 (6), 762–773.
- Strandberg, S., Wretling, M.L., Wredmark, T., Shalabi, A., 2010. Reliability of computed tomography measurements in assessment of thigh muscle cross-sectional area and attenuation. BMC Med. Imaging 10, 18.
- Tarnopolsky, M.A., Pearce, E., Smith, K., Lach, B., 2011. Suction-modified Bergstrom muscle biopsy technique: experience with 13,500 procedures. Muscle Nerve 43, 717–725.
- Tieland, M., Dirks, M.L., van der Zwaluw, N., Verdijk, L.B., van de Rest, O., de Groot, L.C., van Loon, L.J., 2012. Protein supplementation increases muscle mass gain during prolonged resistance-type exercise training in frail elderly people: a randomized, double-blind, placebo-controlled trial. J. Am. Med. Dir. Assoc. 13 (8), 713–719.
- Verdijk, L.B., Koopman, R., Schaart, G., Meijer, K., Savelberg, H.H., van Loon, L.J., 2007. Satellite cell content is specififically reduced in type II skeletal muscle fibers in the elderly. Am. J. Physiol. Endocrinol. Metab. 292, E151–E157.
- Verdijk, L.B., Gleeson, B.G., Jonkers, R.A., Meijer, K., Savelberg, H.H., Dendale, P., van Loon, L.J., 2009a. Skeletal muscle hypertrophy following resistance training is accompanied by a fiber type-specifific increase in satellite cell content in elderly
- men. J. Gerontol. A Biol. Sci. Med. Sci. 64, 332–339.
- Verdijk, L.B., van Loon, L., Meijer, K., Savelberg, H.H., 2009b. One-repetition maximum strength test represents a valid means to assess leg strength in vivo in humans. J. Sports Sci. 27, 59–68.
- Verdijk, L.B., Snijders, T., Beelen, M., Savelberg, H.H., Meijer, K., Kuipers, H., Van Loon, L.J., 2010. Characteristics of muscle fifiber type are predictive of skeletal muscle mass and strength in elderly men. J. Am. Geriatr. Soc. 58, 2069–2075.
- Verdijk, L.B., Dirks, M.L., Snijders, T., Prompers, J.J., Beelen, M., Jonkers, R.A., Thijssen, D.H., Hopman, M.T., Van Loon, L.J., 2012. Reduced satellite cell numbers with spinal cord injury and aging in humans. Med. Sci. Sports Exerc. 44, 2322–2330.
- Wanagat, J., Cao, Z., Pathare, P., Aiken, J.M., 2001. Mitochondrial DNA deletion mutations colocalize with segmental electron transport system abnormalities, muscle fiber atrophy, fiber splitting, and oxidative damage in sarcopenia. FASEB J. 15, 322–332.
- Williamson, D.L., Godard, M.P., Porter, D.A., Costill, D.L., Trappe, S.W., 2000. Progressive resistance training reduces myosin heavy chain coexpression in single muscle fibers from older men. J. Appl. Physiol. 88, 627–633.
- Young, A., Hughes, I., Round, J.M., Edwards, R.H., 1982. The effect of knee injury on the number of muscle fibres in the human quadriceps femoris. Clin. Sci. (Lond.) 62, 227–234.
С уважением, А.В. Самсонова